Painting in 3D: High throughput phenotypic screening in printed 3D cell culture
In collaboration with PerkinElmer (now Revvity).
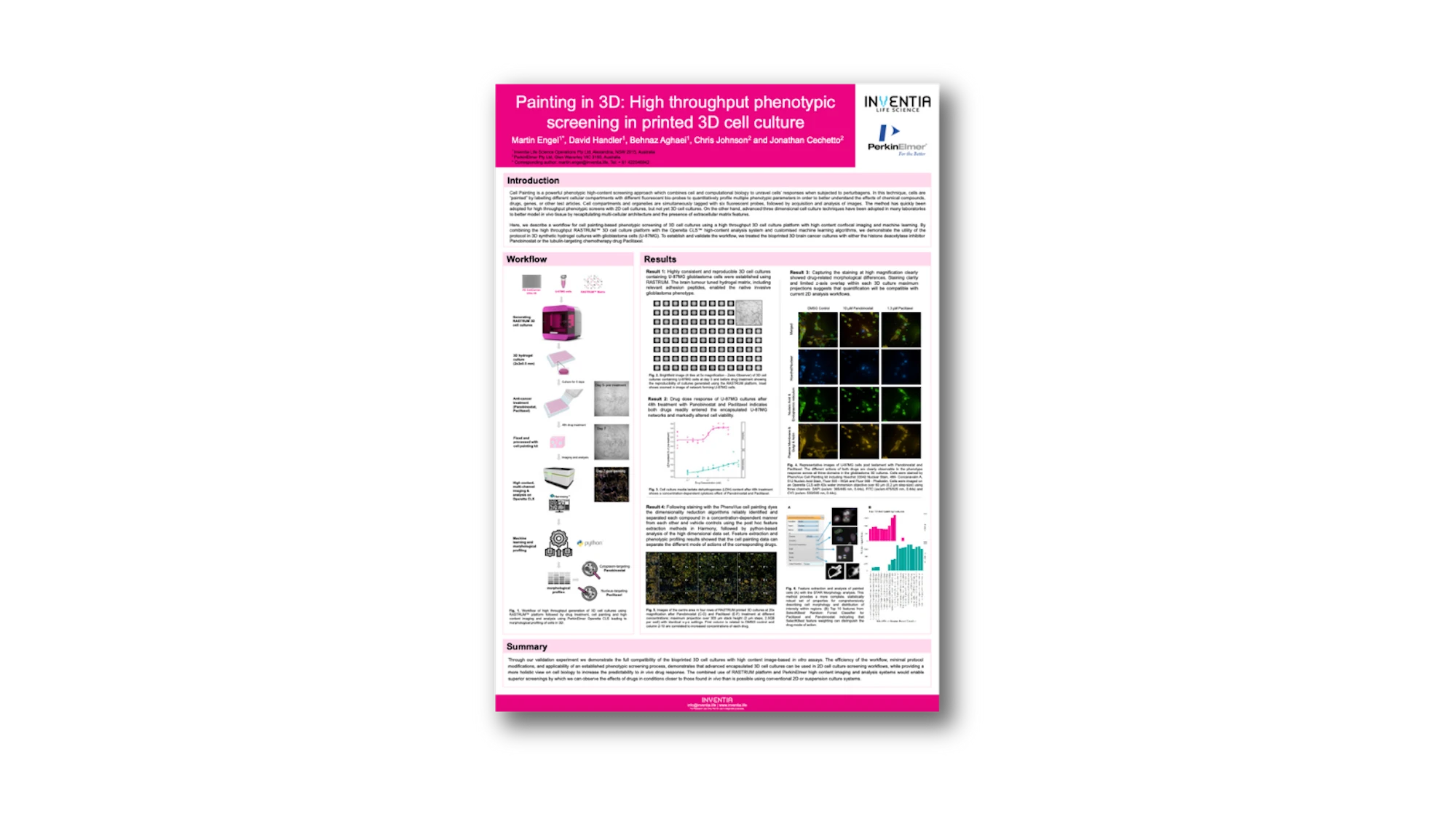
Cell Painting is a powerful phenotypic high-content screening approach which combines cell and computational biology to unravel cells’ responses when subjected to perturbagens. In this technique, cells are “painted” by labelling different cellular compartments with different fluorescent bio-probes to quantitatively profile multiple phenotypic parameters in order to better understand the effects of chemical compounds, drugs, genes, or other test articles.
Abstract
Advanced three dimensional cell culture techniques have been adopted in many laboratories to better model in vivo tissue by recapitulating multi-cellular architecture and the presence of extracellular matrix features. We describe here a workflow for cell painting-based phenotypic screening in 3D cell cultures by using high throughput bioprinting with high content confocal imaging and machine learning. By combining the high throughput bioprinter RASTRUM™ with the Operetta CLS high-content analysis platform and customised machine learning algorithms, we demonstrate the utility of the protocol in 3D synthetic hydrogel cultures with brain cancer (U87MG) cells.
To establish and validate the workflow, we treated the bioprinted 3D brain cancer cultures with either the histone deacetylase inhibitor panobinostat or the tubulin-targeting chemotherapy drug paclitaxel. Both drugs readily entered the encapsulated U87MG networks and markedly altered cell proliferation and morphology. Following staining with the PhenoVue™ cell painting dyes, three-channel 1 mm³ stacks (1.8 x 1.8 x 0.3 mm) were captured at 20x magnification, followed by post hoc feature extraction using the inbuilt Harmony software, and python-based analysis of the high dimensional data set.
The dimensionality reduction algorithms reliably identified and separated each compound in a concentration-dependent manner from each other and vehicle controls. Through our validation experiment we demonstrate the full compatibility of the bioprinted 3D cell cultures with high content image-based in vitro assays. The efficiency of the workflow, minimal protocol modifications, and applicability of an established phenotypic screening process, demonstrates that advanced encapsulated 3D cell cultures can be used in 2D cell culture screening workflows, while providing a more holistic view on cell biology to increase the predictability to in vivo drug response.